 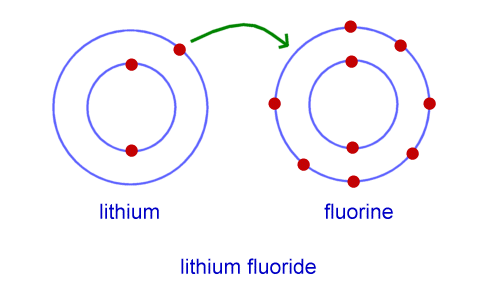
After that, we'll consider the strength of ionic bonding, and ionic radius. 
We'll then explore giant ionic lattices and their properties.We'll define ionic bonding before looking at examples and diagrams of common ionic compounds.This article is about ionic bonding in physical chemistry.Instead, they must gain, lose or share electrons, in order to achieve that optimal configuration. However, other elements don't have this stable arrangement of electrons. This is the most stable electron configuration an atom can have. This is because they all have a full outer shell of electrons. They rarely react with other elements, be it water, oxygen, or metals. Group 8 elements, which you also know as noble gases, are known for being inert. Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.Ion and Atom Photoelectron Spectroscopy.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
